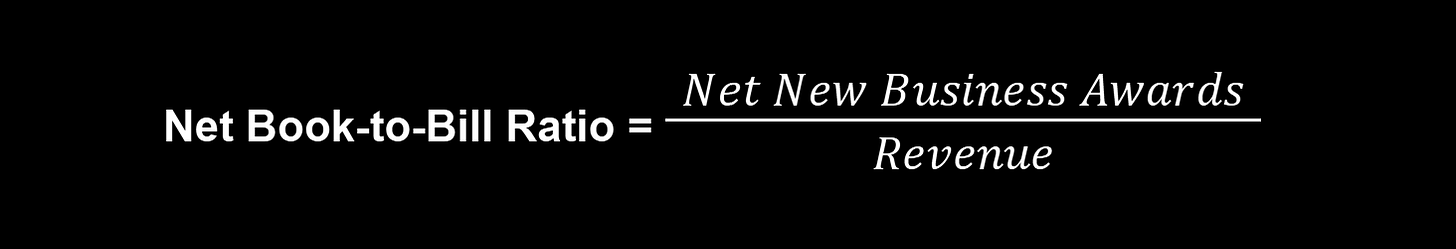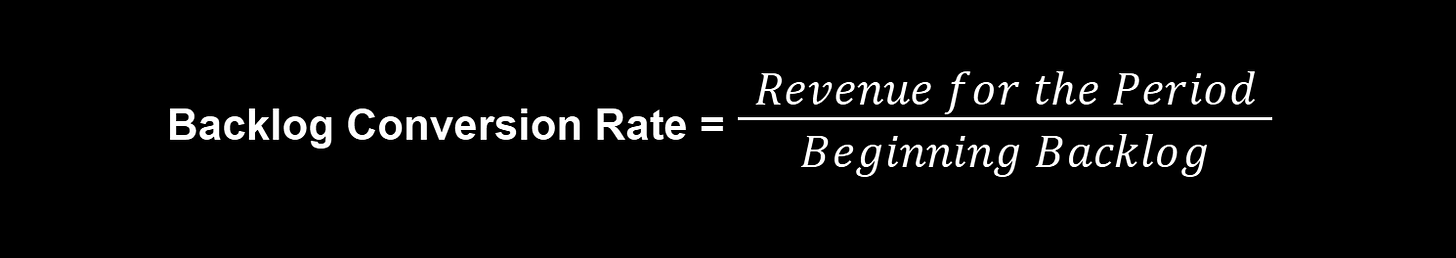The Medpace Multi-Bagger Scenario
Medpace Holdings is one of those companies that I have owned for a while now in small quantity because I wanted to be sure I was at least involved in a small tracker position as I waited for a better opportunity to push in. A recent sell-off related to unforeseen temporary troubles combined with a semi-hidden secular trend that has not yet gained the market’s respect has piqued my interest in a pretty big way. We start with a business that checks all the boxes– founder-led and owned, lots of cash, no debt, a naturally compounding business model, and up-trending margins– and we add in a selloff that makes it attractive as a basement level investment scenario. We then pile on an extremely likely and drastic supercharge of the game that is responsible for funneling Medpace their work in the first place, and suddenly we get a prospect really worth delving into.
I want to walk through the pieces of the puzzle that form the bigger picture of what an investment could look like, starting with a look at who Medpace Holdings is. I think we’ll find that the underlying strength of the business offers a fair base-case opportunity, with a more likely scenario offering a potential multi-bag situation.
The Recurring Revenue Business Model
Medpace Holdings serves the function of bringing drug candidates through the clinical development process. They are not a pharma company themselves, and they do not rely in any way on successful hit rates on newly formulated drugs or winning home run patents. Instead, they take new drugs and do everything that needs to be done, for years and years, to bring them through the end goal of FDA approval. This means that they usher the drugs through all trials and all regulatory processes, which is a multi-year endeavor with no shortage of massive complexities, but from which revenue can be extracted the whole way through. In this way, when Medpace wins a contract, the revenue stream, being multiyear in nature, mimics a recurring revenue model. Every new contract adds on to the already existing revenue streams.
Medpace covers the full lifecycle from early planning through post-approval, often across phase I through IV trials. From beginning to end, they handle everything including:
Planning and design – Protocol development, regulatory strategy, feasibility assessments, etc.
Regulatory affairs – Submitting applications such as IND (Investigational New Drug Application) and CTA (Clinical Trial Application), interactions with agencies like FDA/EMA, and compliance support.
Study start-up – Site selection/activation, contracts, investigator training, and regulatory approvals.
Operational execution – Clinical trial management, monitoring, project management, vendor oversight, patient recruitment/retention strategies, and global site coordination.
Data handling and analysis – Data management, electronic data capture (EDC), biostatistics, pharmacovigilance/safety monitoring, and endpoint adjudication (or Clinical Event Classification (CEC)).
Specialized labs and services – Central laboratories (sample processing/analysis), bioanalytical labs, imaging core lab, ECG/cardiovascular core lab, and Phase I unit for early human studies.
Technology integration – All tied together via Medpace’s proprietary ClinTrak, a single platform for real-time tracking, dashboards, ePRO/eConsent, IRT(Interactive Response Technology) for randomization/supply management, etc.
Close-out and submission support – Database lock, final reports, NDA/BLA preparation assistance, and post-marketing (Phase IV).
Importantly, Medpace classifies themselves as a “full-service” clinical research organization (CRO), meaning they typically would only take on a contract where the full stack of services, from the very beginning planning stages all the way to the very end of approvals and red tape management, are required by the client. The drug does not need to reach FDA approval for Medpace to make a lot of money for a very long time on the contract.
Market Position and an Advantageous Niche
Medpace’s self-classification as a full service CRO is how they differentiate themselves from the competition. Limiting themselves to these types of contracts serves a couple of very important functions.
First, the model attracts smaller players with fewer resources to take on any part of the clinical development process for themselves. Medpace’s bigger sized competitors tend to be less interested in these small players, as they are busy clamoring over huge contracts with the bigger pharma companies of the world. Medpace comfortably operates in this niche of smaller outfits that it has carved out for itself. Bidding wars for contracts are few because of this.
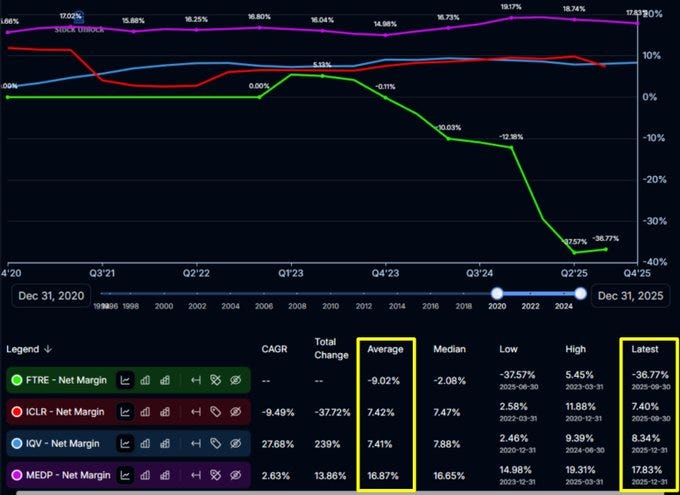
Second, taking on the full stack of the process rather than offering piecemeal, a la carte portions of the process allows Medpace to realize some very beneficial margin benefits over its peers. As the scope of what they do is broader-based, expenses can be stretched further, allowing Medpace to display net margins far above their closest competitors. The full stack service is a competitive, operational advantage for them that shows up in the numbers year after year.
The Lingo
To understand what makes the earnings tick, we have to first get familiar with the interplaying metrics that drive the engine.
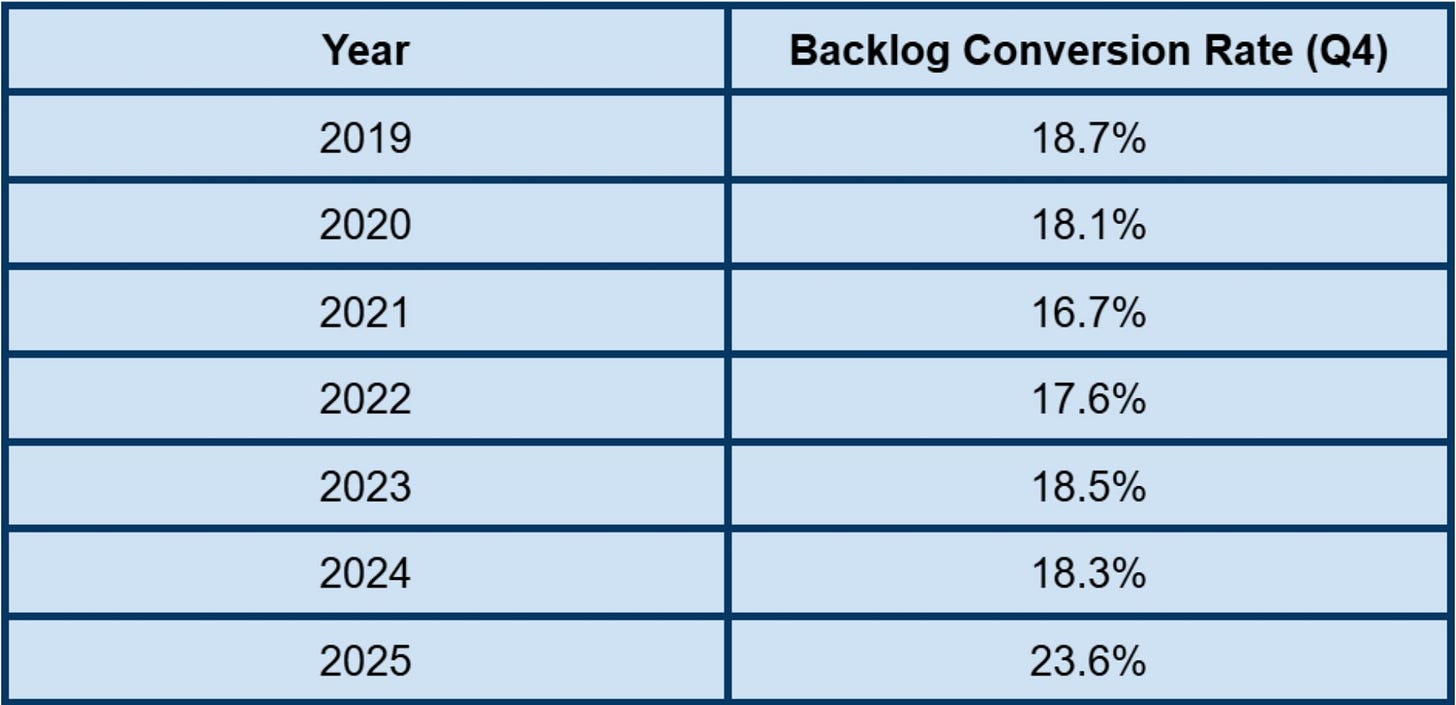
The backlog is an incredibly important metric for Medpace as the entire operation flows to and from that pool of waiting work. Medpace and investors alike track something called the net book-to-bill ratio, which helps everyone monitor how much is flowing into the backlog for every dollar coming out and being realized as revenue when Medpace completes work. Net new business awards ties into the picture as a measurement of new contracts won minus the contracts that were cancelled during the period. Obviously if this number is positive, the pool of backlogged work increases. If it is very, very positive, the backlog can grow substantially.
This brings us to the backlog conversion rate. Medpace measures their success and efficiency by taking the beginning backlog number and measuring how much of it they were able to complete and realize as revenue for the quarter. So, the way we need to think about it is that a huge backlog pool multiplied by a reasonable backlog conversion rate can mean great revenue growth. Or, the flip side is true as well– a reasonable backlog pool multiplied by a very big, efficient conversion rate can also mean good revenue growth. This is arguably what last quarter.
This is why analysts are so focused on the net book-to-bill; they want assurance that the backlog pool is robustly growing so that the conversion rate naturally brings revenue growth. When the book-to-bill falters, analysts get squeamish.
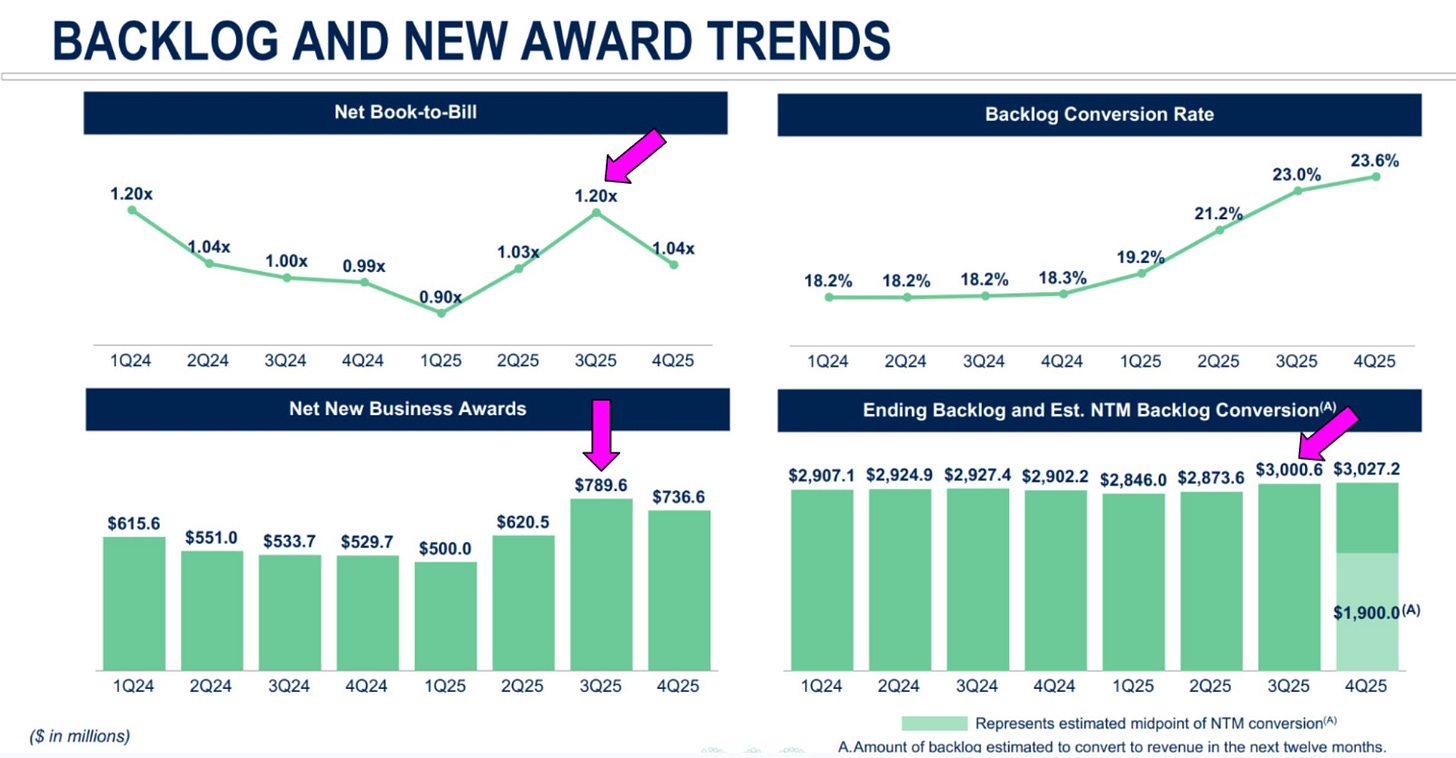
We can see how these metrics interplay in the next image. Note how the book-to-bill and net new business awards move together, and the backlog jumps as the book-to-bill jumps. We can also see how growth of the backlog falters when the book-to-bill drops.
A Compounder on Display
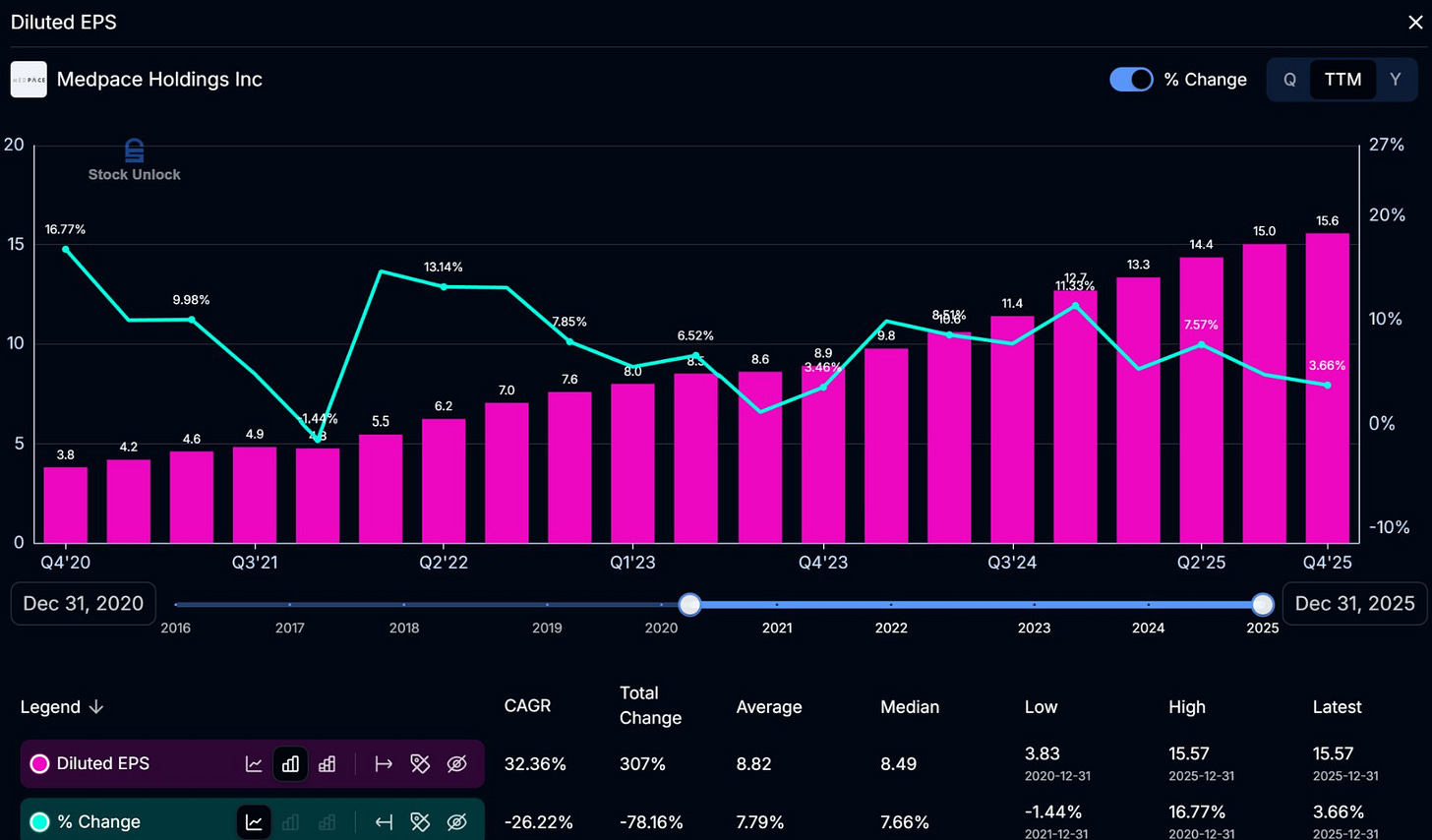
Long-term trends have been strong. This is a robustly compounding earning engine, which is plain to see in the charts.
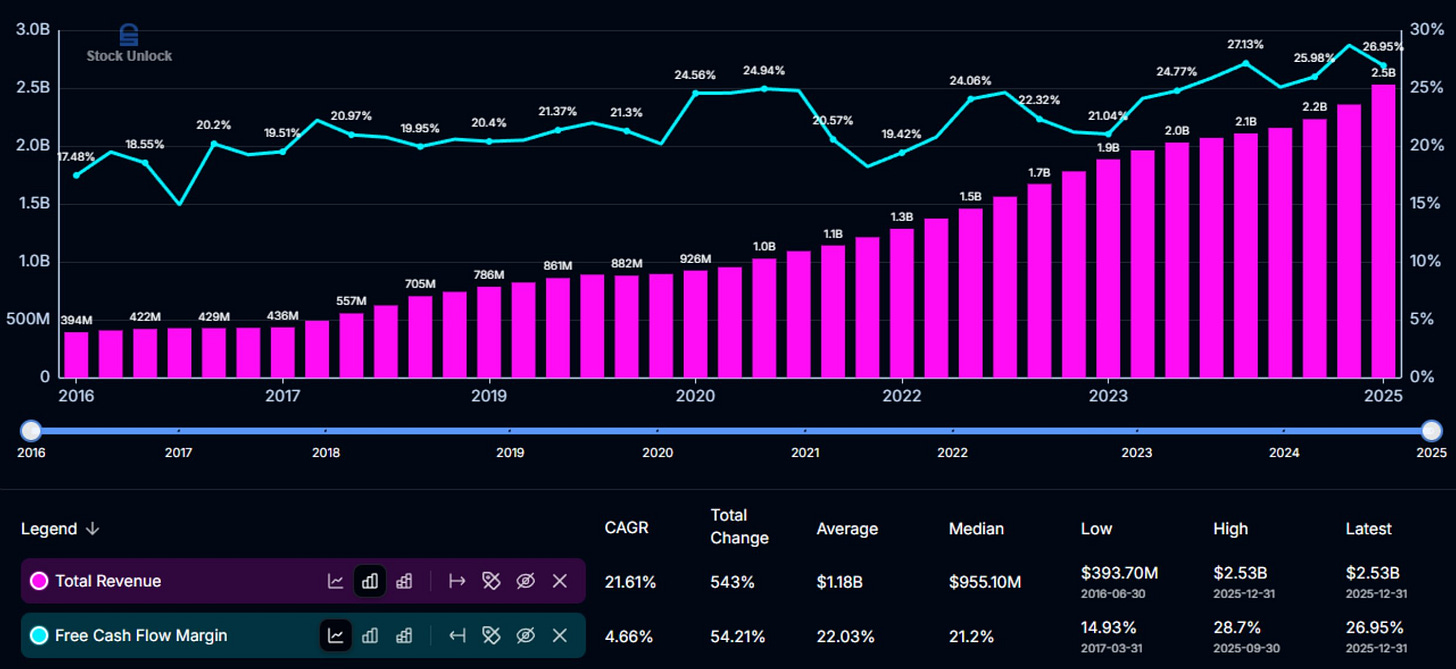
The backlog currently sits around $3B, roughly a quarter the size of Medpace’s entire market cap of ~$12B.
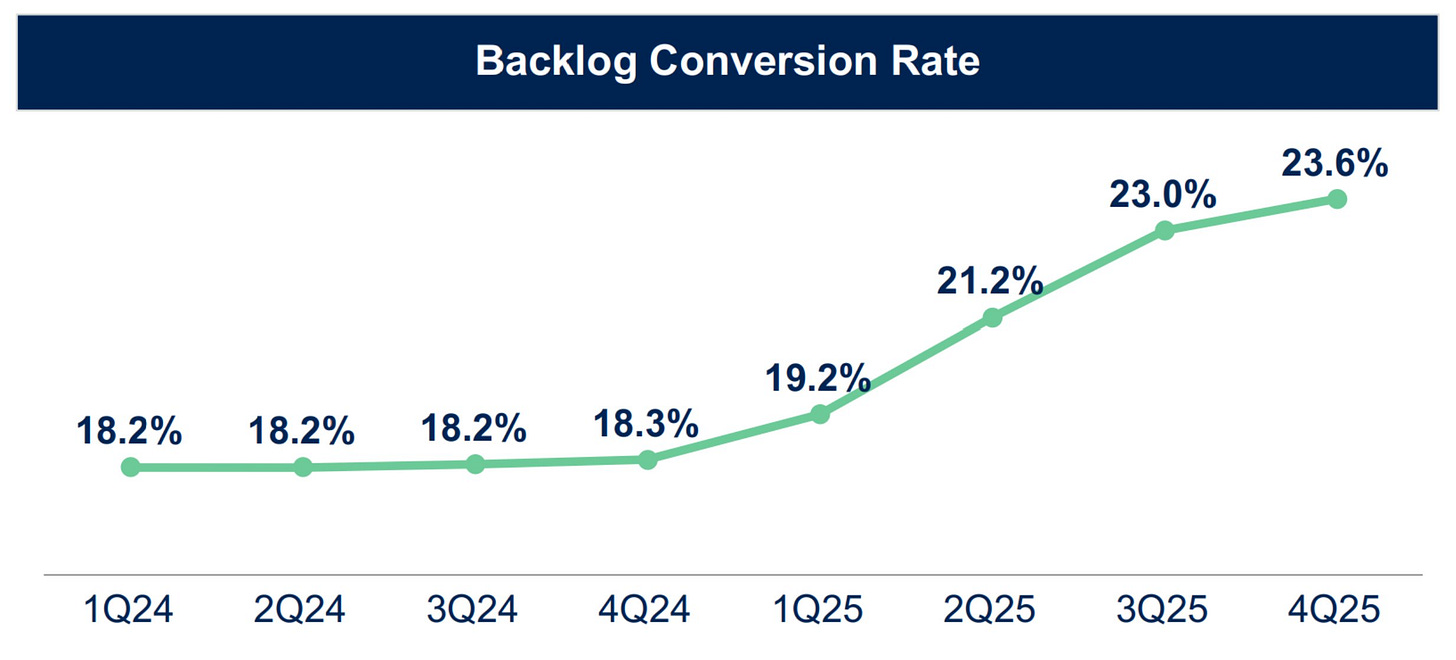
The backlog conversion rate has been on an incline heading north, displaying efficiency in realizing revenue.
The rising backlog conversion rate, while nice to see from an investor’s standpoint, is not guaranteed to stay high. These elevated levels are a reflection of the type of work that is currently being burned through in the backlog; work that is requiring shorter than usual durations leading to more revenue realization in shorter time frames. The type of work that Medpace burns through, however, can shift around, and the backlog conversion rate along with it. Investors should not expect the conversion rates to stay at these levels, and CEO August Troendle has said as much.
Recent Selloff
In the latest earnings report, Q4 of 2025, Medpace reported a surprise jump in contract cancellations that nobody, including management, saw coming. The cancellations negatively affected the net new business awards, which translated to a book-to-bill ratio of 1.04, far lower than had previously been reported in recent quarters. When asked by analysts about the nature of the cancellations, Troendle said that there was no discernable pattern or reason that they could identify, and that the cancellations were of a broad-based nature for all kinds of unrelated reasons that were specific to each company involved. It essentially felt like an unlucky fluke. He had no reason to believe we would see cancellations like this again next quarter, as there was nothing macro economic or industry-related in play here. This framing of the cancellation issue as a transient problem will be key to our thesis and the way we view the valuation.
The market saw the falling book-to-bill and the resulting relative stagnation of the backlog, and took it as a sign that earnings growth could be challenged moving forward. The stock sold off 15 to 20%, potentially offering us a contrarian opportunity.
When we combine time with an ability to label a poor development, or series of poor developments, as impermanent, that is the equation for safe contrarianism.
– David Perlmutter, 8th Wonder Investment
Interestingly, the company displayed strong earnings growth for both the quarter and year off the back of a very strong backlog conversion rate, as previously mentioned. As previously stated, for earnings to grow, either the backlog has to have good growth or the conversion rate has to be high. Since we can’t count on the conversion rate staying as high as it is due to shifts in the type of work that Medpace is tackling at the time, investors need to know that the book-to-bill will continue to feed the backlog. With the cancellations of Q4 2025 likely being an anomaly, I see no reason to fear it as a new trend. This happens from time to time in Medpace’s world, and they always recover to more normalized growth trends.
Important note:
Regarding the relationship between backlog conversion rate and the backlog, it is vitally important to understand that as the conversion rates are higher, it makes it harder for the backlog to grow as work is being funneled from it faster than usual. Ironically, Medpace’s efficiency can cannibalize the backlog. For example, in the most recent quarter the backlog had only grown by around 4% Y/Y. Net new business awards were $736.6M but the conversion rate took $708.14M from the pool. If the conversion rate had been 18%, which is more inline with historical averages, $540.18M would have been pulled from the backlog instead which would have meant that the backlog would have seen growth of $196.42M, or 6.5% in just this quarter alone. This also would have represented a Y/Y backlog growth rate of 10.2%. This is true in the face of an unprecedented level of cancellations, meaning that, had cancellations been more along an average level, the backlog growth would have been far more robust.
The recent high backlog conversion rate, along with some well-placed buybacks, have kept the growth story intact as the backlog faltered from the flare-up in cancellations.
A Ground-Floor Base Case
Before moving on to the explanation of how we could enter multi-bagger territory with this stock, we can lay the foundation for a “tails, I don’t lose much” investment case. We can start by showing what what we could reasonably expect if the earnings growth rate meaningfully retracts. To show these simply, we can use a pricing model, complete with a discount rate, to estimate returns and gauge under or over valuation. Stock Unlock’s DCF calculator is utilized here for its clear depiction of the investment scenario and ease of use.
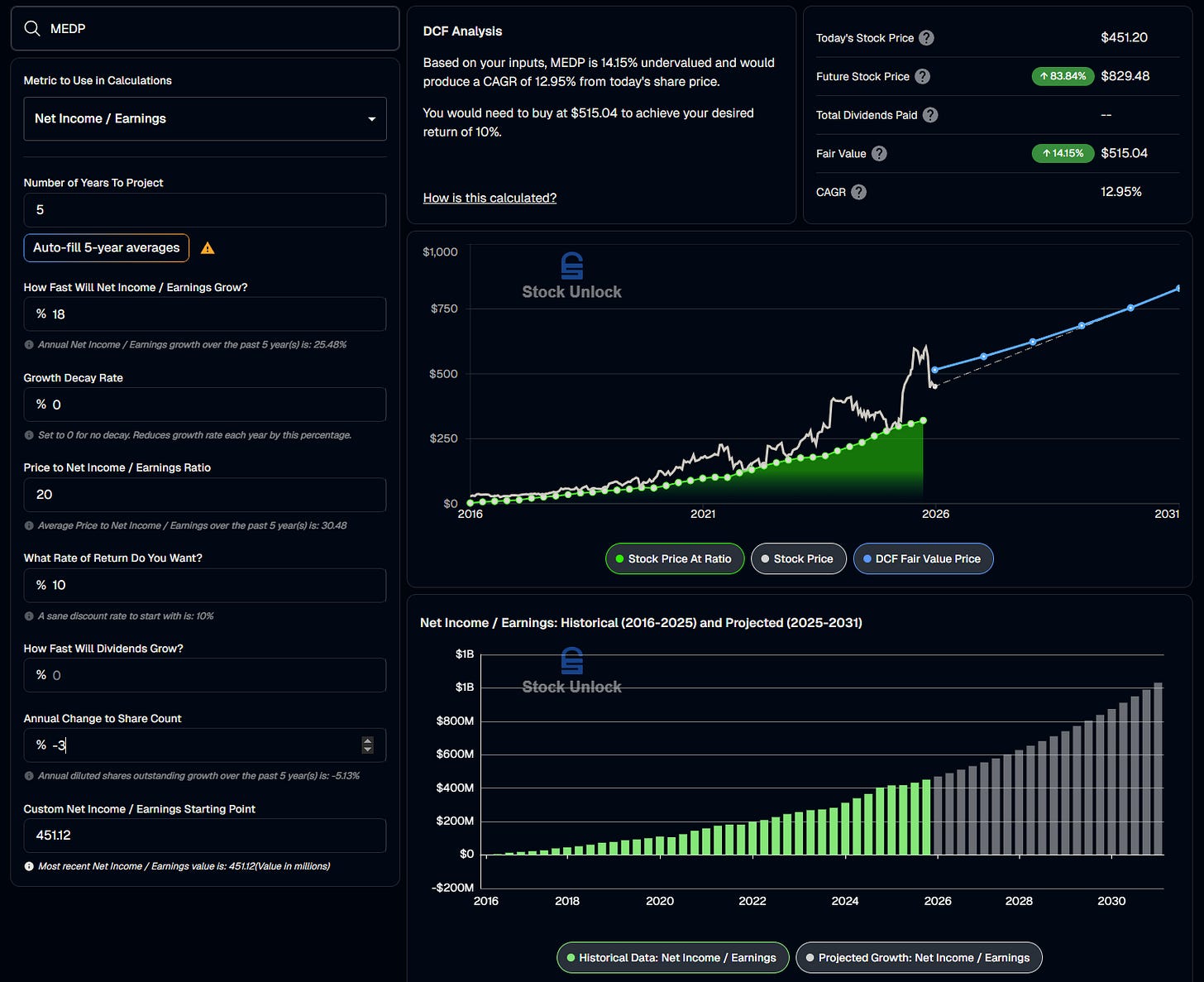
The 5 year average earnings growth rate is 25.48%.
If we assume a marked decline in earnings growth rate to 18% on average over 5 years, and bring the P/E down by 50% from an average of 30.48 to 20, we can expect a return of around 13% per year on a purchase today.
This serves as a good base case before we even get into a discussion about the paradigm that will likely amplify returns profoundly down the line. To understand the rest of the picture, we have to explore the changing landscape within the sector. The pluses and minuses of its evolution are both important to review.
Evolution of the FDA Regulatory Landscape
In December 2025, FDA Commissioner Marty Makary announced plans to shift the default requirement from two pivotal clinical trials (phase III) to one for many new drugs and medical products. A phase III trial is a source of work for anywhere from 1 to 5 years, with most taking around 2 years from beginning to end for Medpace specifically. Naturally, as the market understood that the announcement looks to shave off years of work from CROs’ trialing contracts, these stocks sold off.
However, as in all things, we have to take it in a broader context, especially as it relates specifically to Medpace. It is helpful to remember that Medpace purposefully limits themselves to full stack contracts. They are a “full service” CRO, with their contracts spanning the entire clinical development process from beginning to end. With the full breadth of services typically lasting anywhere from 5 to 10+ years, the elimination of a single phase III study from the stack of services still results in typical timespans that will average around the half decade mark. Though some work is undoubtedly lost to the FDA’s new focus on streamlining the process, Medpace is somewhat uniquely shielded from the effects due to the wide span of their service commitments. It remains true that everything in the backlog still represents years and years of recurring revenue. The compounding model remains intact as long as they can continue to grow the backlog, just as they always have.
Evolution of the Drug Discovery Process
This brings us to AI, and the new drug discovery cadence that is emerging. Deepmind’s Alphafold was an incredible breakthrough, to say the least. Demis Hassabis’s Nobel Prize was very well deserved as he brought to the whole world the ability to solve the long-standing protein folding problem – figuring out how a chain of amino acids, the sequence of a protein, folds into its functional 3D shape. This shape determines what the protein does in the body, which is why it is so important to understand for drug discovery.
But Demis Hassabis took it a step further when he spun out a company called Isomorphic from Deepmind. Isomorphic’s goal is to bring something even more valuable to the world. They are focused on creating an AI-powered drug design engine that models complex biological systems, predicts molecular structures and interactions, and designs novel drug candidates. The effect of a working system like this is that the entire drug discovery process wildly accelerates, as much of the work moves from the clunky, inefficient wet lab to the fast and efficient world of the in silico. What took years or decades to accomplish in bringing forth a new drug candidate will now take months or weeks.
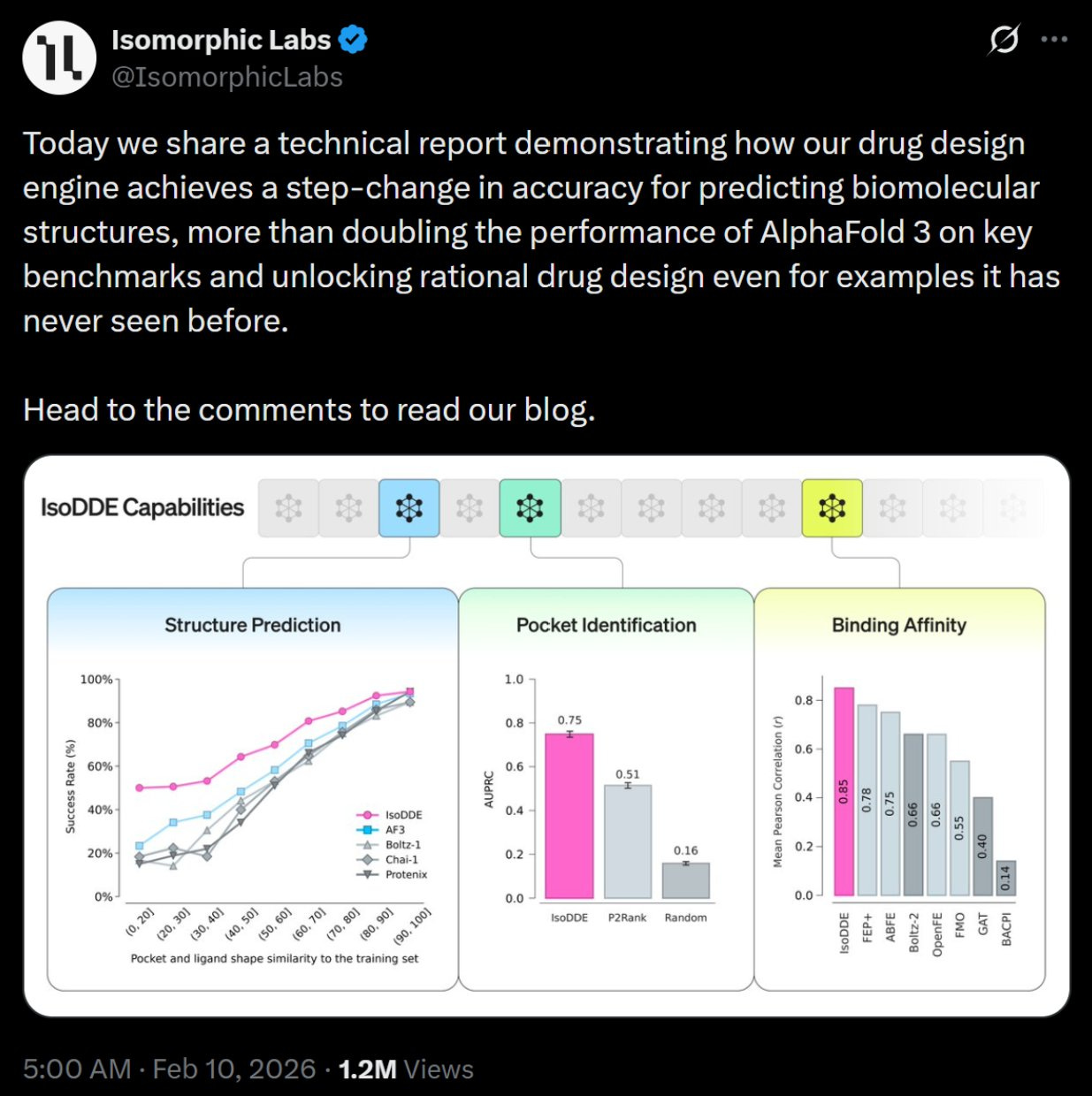
This sounds like some far off science fiction, something for us to be hopeful for achieving sometime down the line. But, actual real progress has been made; the kind that is useful in the immediate present. Just this month (February, 2026) Isomorphic released details on its drug design engine IsoDDE, which significantly outperforms AlphaFold 3 on key benchmarks and more than doubles accuracy on difficult protein-ligand structures, improves generalization to novel systems, and enables rational drug design for unseen cases.

These advancements have led to an internal pipeline of AI-designed drug candidates, with multiple programs now at the preclinical stage. Hassabis has said that the first AI-designed drugs from Isomorphic are expected to enter human clinical trials by the end of 2026. Let that sink in for a moment.
Naturally, as more of the drug discovery process moves from the wet lab to in silico, time of synthesis drops while accuracy rises. Both of these shifts reduce costs and slash project funding risks. The real takeaway here is that there is an incoming environment where funding is more readily available, translating to more overall projects, and hit rates on those projects are higher. These effects are poised to compound into a rapid escalation in available CRO contracts as every new and upcoming drug will need to be brought through the full stack of the clinical development process.
Furthermore, in silico advancements and decreased funding risk will mean falling barriers to entry for smaller, more risk-averse would-be drug discoverers. We are likely to see more work coming from smaller players with fewer resources. This uniquely benefits Medpace, far more than any other CRO, since they are a full-service partner with outsized value to smaller players who have no ability to take on parts of the clinical development process themselves. In fact, these types of clients account for about 85% of Medpace’s work today, as is their design. Medpace has always catered to these players, and now this cohort of drug discoverers, with softer barriers to entry and less risk, are set to multiply in number.
Finally, higher reliability in drug design with higher hit rates and easier funding will send CRO contract cancellations plummeting. Cancellations have always been a thorn in Medpace’s side, and now two of the biggest culprits for causing them are going by the wayside. This should usher Medpace’s book-to-bill ratio higher while keeping it more consistent.
So, to summarize, we are set to see an influx of smaller drug discoverers from easing barriers to entry (think projects coming out of universities and the like), higher hit rates on drug discovery projects, shorter time-frames in the drug discovery process, and an easier, less risky funding environment coming as a result of all of this put together. The number of available CRO contracts will soar while cancellations dive. And Medpace is perfectly positioned to absorb the benefits.
The question for our multi-bagger thesis will ultimately be the following:
In a world where the drug discovery process moves from taking years to mere months, likely a ten fold increase in speed, while cancellations drop due to increased accuracies, how will the backlog number react? How will the net-book-to-bill react?
Again, these questions are not based on some far off science fiction. In silico advancements that have already taken place are bringing real new drugs to human trials presently.
If speed doubles, the backlog will do well. If speed moves 10x, the backlog will be absolutely drowning in new work. Apply any backlog conversion rate you want to a backlog affected this way, and it becomes easy to see how the stock price could multiply to keep up with the earnings growth.
Understanding with accuracy the sheer impact of an environment like this on Medpace’s future backlog would be challenging, to say the least. However, we can frame the idea of a possible investment in MEDP as having the base case scenario initially shown above, with the new incoming AI in silico landscape potentially acting as a massive call option for the position. If the investment lands anywhere along this spectrum, it should work out extremely well.
For this thesis, we may have to augment the famous quote:
“Heads, I win big. Tails, I likely still win.”
Not financial advice.